PDF chapter test TRY NOW
Characteristics of chemical changes:
- A new substance is formed which have a different chemical composition from that of the reactants.
- It is a permanent change.
- It is irreversible.
- Heat, light, and sound is evolved during change. In some cases, it may be absorbed.
Conditions needed for a chemical change:
1. Pressure:

Pressure cooker
Cooking rice grains in a pressure cooker under pressure causes changes in rice grains. This change in texture and taste of the cooked rice is an example of chemical change due to pressure.
2. Heat:

Incense stick
The burning of incense stick changes into ash which is an example of chemical change due to heat.
3. Electricity:
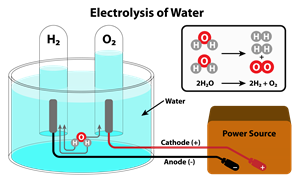
Electrolysis reaction
The electrolysis reaction where electricity is passed causes a new substance to be formed. For example, the electrolysis of water (liquid) produces hydrogen (gas) and oxygen (gas).
4. Physical contact:

Woods in contact with fire
For chemical reaction or chemical change to occur, we need the substance to be physically in contact. For instance, a chemical change to occur in wood, it needs be in contact with fire.
5. Light:
5. Light:
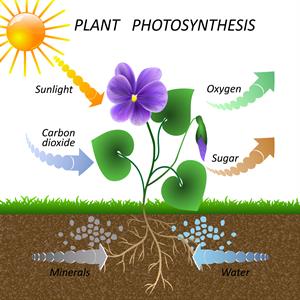
Photosynthesis
Chemical changes occur with the help of light. For instance, the process of photosynthesis takes place with the help of light. In this process, glucose and oxygen are produced.
6. Bubbles:
6. Bubbles:

Bubbles formation
The reaction of sodium bicarbonate with lemon extract is a chemical change. The product formed is sodium citrate, a salt with a different chemical composition than that of the reactants. The \(CO_2\) gas bubbles are evolved with the hissing sound during this reaction.
Indicators of a chemical change:
1. Colour change:
1. Colour change:

Colour change
Some of the chemical changes also occur with colour change. The ripening of the banana is an example of chemical change as the raw banana is green in colour. Incontrast, the ripened banana is yellow and also, the chemical composition of the ripened banana is different from that of the raw banana.
2. Temperature change:

(a)Exothermic reaction (b)Endothermic reaction
Burning of wood produces ashes which are accompanied by heat and light. This is an example of an exothermic reaction as heat is liberated. Whereas, photosynthesis is an endothermic reaction as energy is absorbed from the surroundings.
3. Light and heat are produced:

Firecrackers
The bursting of firecrackers is an example of light and heat produced during the reaction.
4. Odor and taste changes:

Foul smell

Baked cookies smell
The smell and taste of spoiled food and baked foods are examples of chemical changes indicated by smell.
5. Precipitate formation and Volume Change:

Bubbles and precipitate formation
The bubbles formed during the reaction of sodium bicarbonate with lemon extract causes rapid expansion in the reactants' volume. The precipitate formed settles at the bottom of the test tube. This is an example of a chemical change that occurs with a change in volume and precipitate formation.
