PDF chapter test TRY NOW
Chemical Formulae:
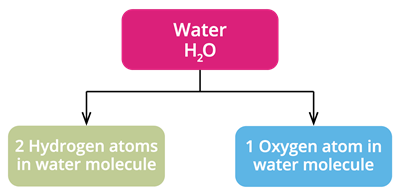
We know water and the molecule's chemical formula as \(H_2O\). It means that each water molecule contains two hydrogen atoms and one oxygen atom combined.
A chemical formula is a description of one molecule of an element or compound in symbolic form. It indicates how many atoms each element has and how many elements are in the molecule.
The chemical formula tells us how many of each kind of atom are there are in one molecule of a substance and all other information required.
Note: Subscript refers to a small number next to the symbol \(X_2R\). Subscript indicates how many atoms of that element are present in the molecule.
Now, let us take few molecules as examples:
1. Water
Chemical formula: \(H_2O\)
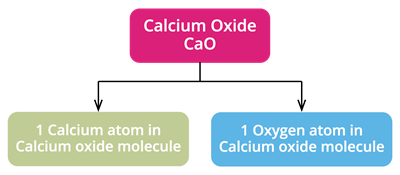
2. Calcium Oxide
Chemical formula: \(CaO\)
3. Glucose
Chemical formula: \(C_6H_{12}O_6\)

In the above examples, chemical names such as water, glucose, and calcium oxide are called compounds. These can be separated further into their elements through chemical methods.
To remember:
- The compounds are substances that have fixed components and can be broken down into elements through chemical reactions.
- The substances which cannot be broken down to the simpler form of substances is known as the elements.
