PDF chapter test TRY NOW
Balancing acids and bases is important for our health and our environment. We come across various neutralisation reactions in our daily life. Let us study the importance of some of these reactions.
Bee bite
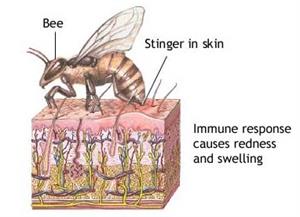
Bee bite
Bees and red ants both contain formic acid. When bees or red ants bite us, they send the acid into our bodies. This acid creates pain and a burning sensation. To neutralise the formic acid and reduce the pain, a suitable base in the form of calcium hydroxide (lime paste available at home) is applied.
Wasp bite

Wasp bite
When a wasp bites, it injects an alkaline substance into our bodies, causing pain and a burning sensation. To neutralise the alkalinity, we add vinegar, which is an acid.
Tooth decay

Decay of tooth
Doctors generally recommend that we wash our teeth twice a day. This is because the bacteria in our mouth decompose the food particles trapped between our teeth, resulting in the development of acid, which causes tooth decay. To prevent this, we have to neutralise the acid. So when we brush with tooth powder or toothpaste containing weak bases, the acid gets neutralised. As a result, we will have healthy and strong teeth.
Acidity
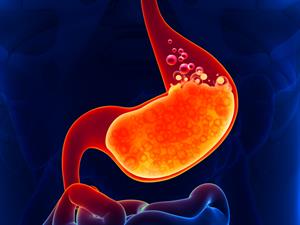
Stomach acidity
As we all know, the hydrochloric acid in our stomach helps in the digestion of food. Due to the excessive production of hydrochloric acid in our stomach, we feel a burning sensation in the food pipe and the chest area. If it prolongs for a long time, the ulcer will be formed in the stomach and food pipe, which further aggravates the conditions. In that case, antacids are used to neutralize. Antacids are weak bases like aluminium and magnesium hydroxides.
Agriculture
In agriculture, the nature of the soil is important. Plants do not grow well in acidic soil. To neutralise the acidity, add lime fertilisers such as powdered lime (\(CaO\)), limestone (\(CaCO_3\)) or burnt wood ashes to the soil.
Industries

Industry waste
Acids such as sulfuric acid can be found in industrial effluents. Before being discharged into rivers and streams, it is treated by adding lime to neutralise it. Similarly, coal and other fossil fuels are burned to generate electricity in power plants. When fossil fuels are burned, sulphur dioxide gas is released into the atmosphere as an acidic pollutant. As a result, power plants neutralise this acidic gas with powdered lime (\(CaO\)) or limestone (\(CaCO_3\)) to prevent air pollution.
Reference:
https://media.istockphoto.com/photos/striped-angry-wasp-stuck-a-sharp-thorn-in-the-human-skin-picture-id842606604
https://biz.libretexts.org/@api/deki/files/20840/Clean-Water-Act-Image.jpg?revision=1
http://i.stack.imgur.com/oNIH8.jpg
