
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoEverything in the Universe is made up of atoms and molecules.
Atoms are mainly composed of particles such as electrons, protons, and neutrons. In an atom, the negatively charged particles are the electrons, the positively charged particles are the protons, and the chargeless particles are the neutrons.
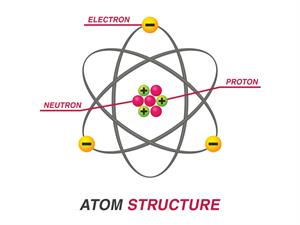
An atom
The nucleus of an atom:
An atom consists of a nucleus with positively charged protons and chargeless neutrons. Also, the negatively charged electrons orbit the nucleus. The number of electrons in an atom is usually equal to the number of protons, thus making every atom neutral in itself.
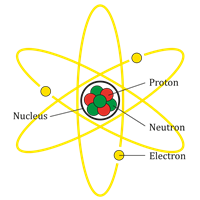
Nucleus of an atom
Ion:
An ion is formed when an atom either receives or loses electrons.
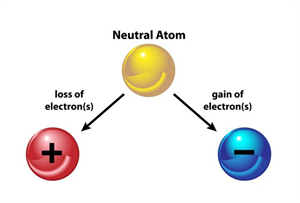
Ion
Positive ion:
If an electron is removed from the atom, the atom becomes positively charged. Then, it is called a positive ion.
An atom with an equal number of protons and electrons has no charge. However, if a negatively charged electron is removed, the atom will have more protons and thus becomes a positive ion.
Negative ion:
If an electron is added in excess to an atom, then the atom is negatively charged, and it is called a negative ion.
Try this out!
Take a plastic comb and rub it over your dry hair. Now, place the comb near small bits of paper and observe.
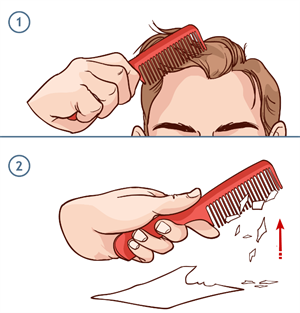
Attraction of paper bits to the comb
It is seen that the plastic comb attracts those small pieces of paper when placed near to it.
Electrons present in your hair leaves and gets collected on the comb's edge when you rub it vigorously. Now, your hair is positively charged as electrons have been lost, whereas the comb is negatively charged as electrons have been gained.
Reference:
https://upload.wikimedia.org/wikipedia/commons/thumb/8/80/Atom_Diagram.svg/1200px-Atom_Diagram.svg.png